Nanotube Blockade May Slow Huntington’s Spread
Nanotube Blockade May Slow Huntington’s Spread📷 Published: Apr 16, 2026 at 04:32 UTC
- ★Tunneling nanotubes transfer mutant protein
- ★Study suggests targeting nanotubes reduces spread
- ★No immediate clinical application yet
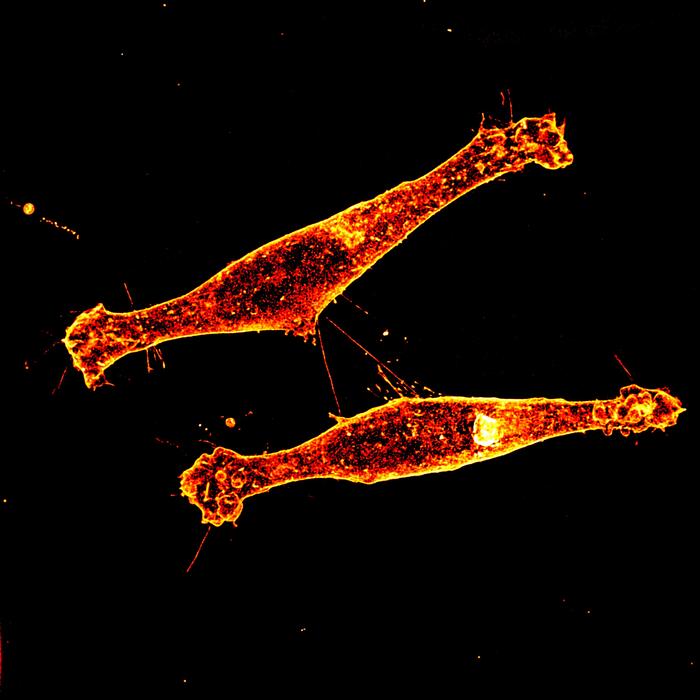
Huntington’s disease has long eluded effective treatment, but a new study points to a potential chink in its armor: tunneling nanotubes. These microscopic bridges between brain cells allow the mutant huntingtin protein—the root cause of the disease—to spread from cell to cell, accelerating neurodegeneration. Researchers at GEN News have now identified a cellular pathway that facilitates this transfer, offering a tantalizing target for future therapies.
The findings, published in Genetic Engineering and Biotechnology News, confirm that disrupting these nanotubes can reduce the intercellular transmission of the pathogenic protein. This isn’t the first time tunneling nanotubes have been implicated in disease progression—similar mechanisms have been observed in cancer and Alzheimer’s—but the specificity to Huntington’s adds a new layer of urgency. The study’s evidence, while compelling, is still preclinical, meaning it was conducted in laboratory models rather than human patients.
For now, the work remains firmly in the realm of basic science. The researchers used experimental interventions, such as inhibitors or genetic modifications, to block nanotube formation, but these methods are far from ready for clinical trials. The mechanism itself—how exactly the mutant protein hijacks these cellular highways—is still being unraveled. What’s clear is that this pathway exists, and targeting it could, in theory, slow the disease’s spread.
Early research reveals a cellular pathway—but patient impact remains distant📷 Published: Apr 16, 2026 at 04:32 UTC
Early research reveals a cellular pathway—but patient impact remains distant
The study’s limitations are worth noting. Sample sizes were small, and the experiments were conducted in controlled environments that don’t fully replicate the complexity of the human brain. While the results are promising, they fall short of proving that nanotube disruption will work in patients. Clinical relevance remains speculative at this stage, with no guarantee that blocking these nanotubes will translate into tangible benefits for those living with Huntington’s.
Regulatory hurdles add another layer of uncertainty. Even if future research validates these findings, developing a safe and effective therapy will take years. The FDA’s approval pipeline for neurodegenerative diseases is notoriously slow, with many promising candidates failing in late-stage trials. For patients and families desperate for progress, this study offers a glimmer of hope—but not a cure.
What we still don’t know looms large. Does blocking nanotubes have unintended consequences for healthy brain function? Could other pathways compensate for the loss of this one? And most critically, will this approach work in humans, or is it another dead end in the long hunt for a Huntington’s treatment? The answers will require more research, more funding, and more time—resources that are never guaranteed in the competitive world of biomedical science.
In other words, this study doesn’t prove a therapy, but it does reveal a vulnerability in Huntington’s disease that wasn’t fully understood before. The mutant protein’s spread isn’t inevitable—it relies on a specific cellular pathway that can, at least in principle, be disrupted. That’s a far cry from a clinical breakthrough, but it’s a step toward one.