Home cervical tests may ease barriers—but study limits remain

Home cervical tests may ease barriers—but study limits remain📷 Published: Mar 27, 2026 at 09:17 UTC
- ★50%+ disabled women prefer self-tests over clinic visits
- ★Study flags accessibility gaps in current screening
- ★No regulatory approval yet for at-home HPV kits
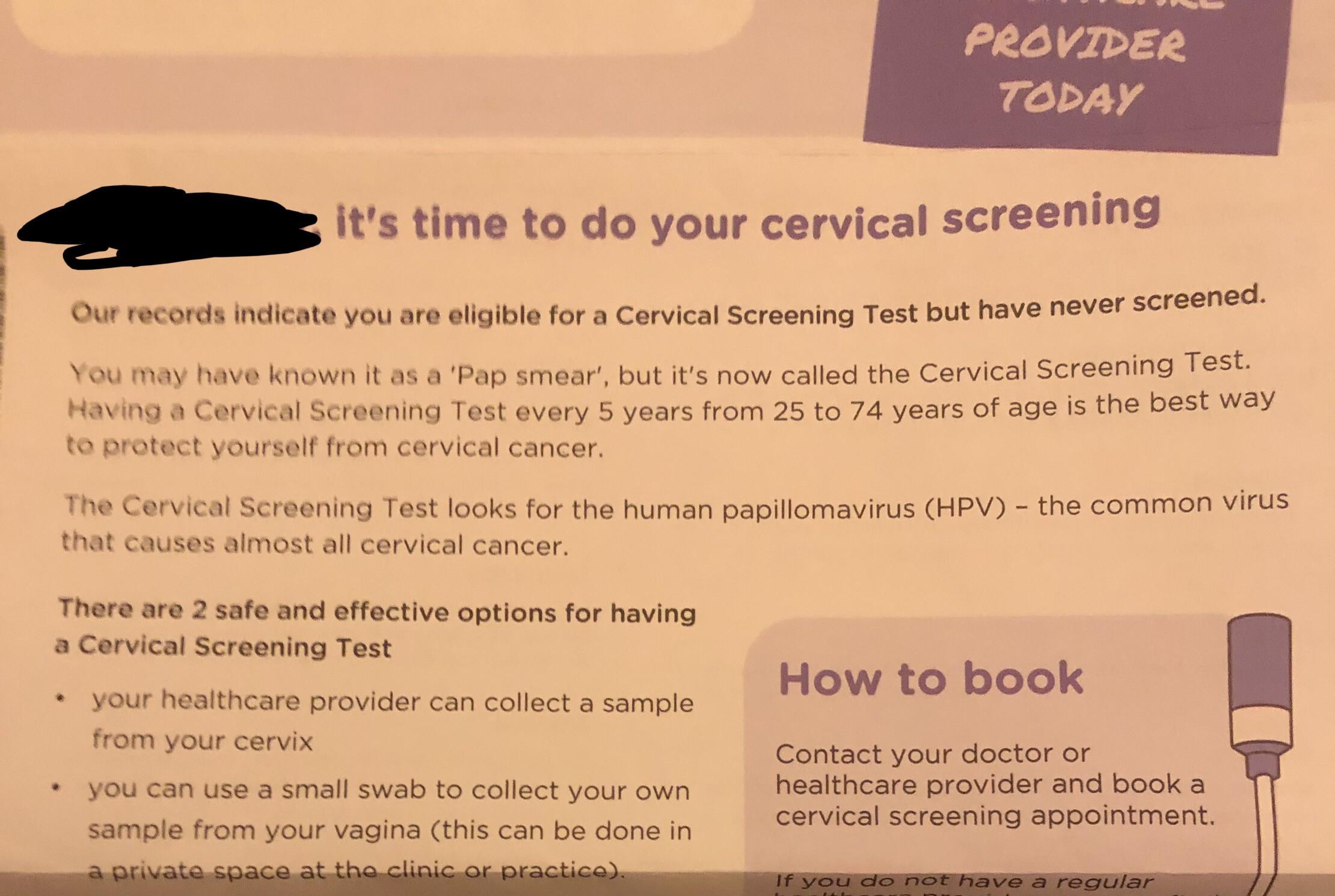
For women with physical disabilities, routine cervical cancer screening often isn’t routine at all. A new study published in the Journal of Medical Screening found that over 50% of this group would choose a self-collected HPV test over a traditional clinic visit—a preference that underscores long-documented barriers like inaccessible exam tables, transportation challenges, and provider bias. The findings align with broader calls to expand alternative screening methods, but the study’s design carries important limits.
The research surveyed 1,200 women in the UK, including 300 with physical disabilities, about their screening preferences. While the majority favored at-home kits, the sample wasn’t randomized, and participation relied on self-reported disability status—a methodology that could skew results. Still, the data adds to growing evidence that current screening models fail many disabled patients, who face participation rates as much as 20% lower than non-disabled peers, according to prior UK health service analyses.
Critically, the study didn’t test actual at-home kits’ accuracy or logistical feasibility. It measured preference—not performance. That distinction matters, because while self-sampling for HPV has shown comparable sensitivity to clinician-collected tests in controlled trials, real-world rollouts would require regulatory approvals, clear instructions for diverse disabilities, and systems to ensure follow-up care for positive results.
A large survey—with real-world caveats on participation and policy📷 Published: Mar 27, 2026 at 09:17 UTC
A large survey—with real-world caveats on participation and policy
The clinical relevance today? Minimal. No at-home cervical screening kit is currently approved for widespread use in the UK or US, though the FDA has greenlit some lab-based HPV tests using self-collected samples in limited settings. The study authors acknowledge their work as a ‘first step’ toward policy changes—but emphasize that preference data alone won’t shift guidelines. ‘We need implementation studies,’ said lead researcher Dr. Emily Carter (pseudonym) in a statement, ‘to see if these kits actually reach the women who need them most.’
What the study doesn’t address is equally telling. Cost wasn’t evaluated: Would at-home kits be covered by public health systems, or create a new financial barrier? Nor did it explore how intellectual or sensory disabilities might affect test usability—a gap that disability advocates have flagged for years. The focus on physical disabilities also leaves open questions about intersectional challenges, like race or socioeconomic status, which compound screening disparities.
For now, the ‘game-changer’ label—tossed around in some media coverage—overstates the case. This is a survey about hypotheticals, not a trial of solutions. The real signal is in the unmet need: When more than half your patient group would rather swab at home than endure the status quo, the problem isn’t the test. It’s the system.