
Dr. Donald Kohn's hands, warm skin tones, holding a petri dish with a gene therapy sample, minimal vector concept art, two-tone palette of clean📷 Photo by Tech&Space
- ★Gene therapy approved
- ★Severe leukocyte adhesion
- ★Dr. Donald Kohn
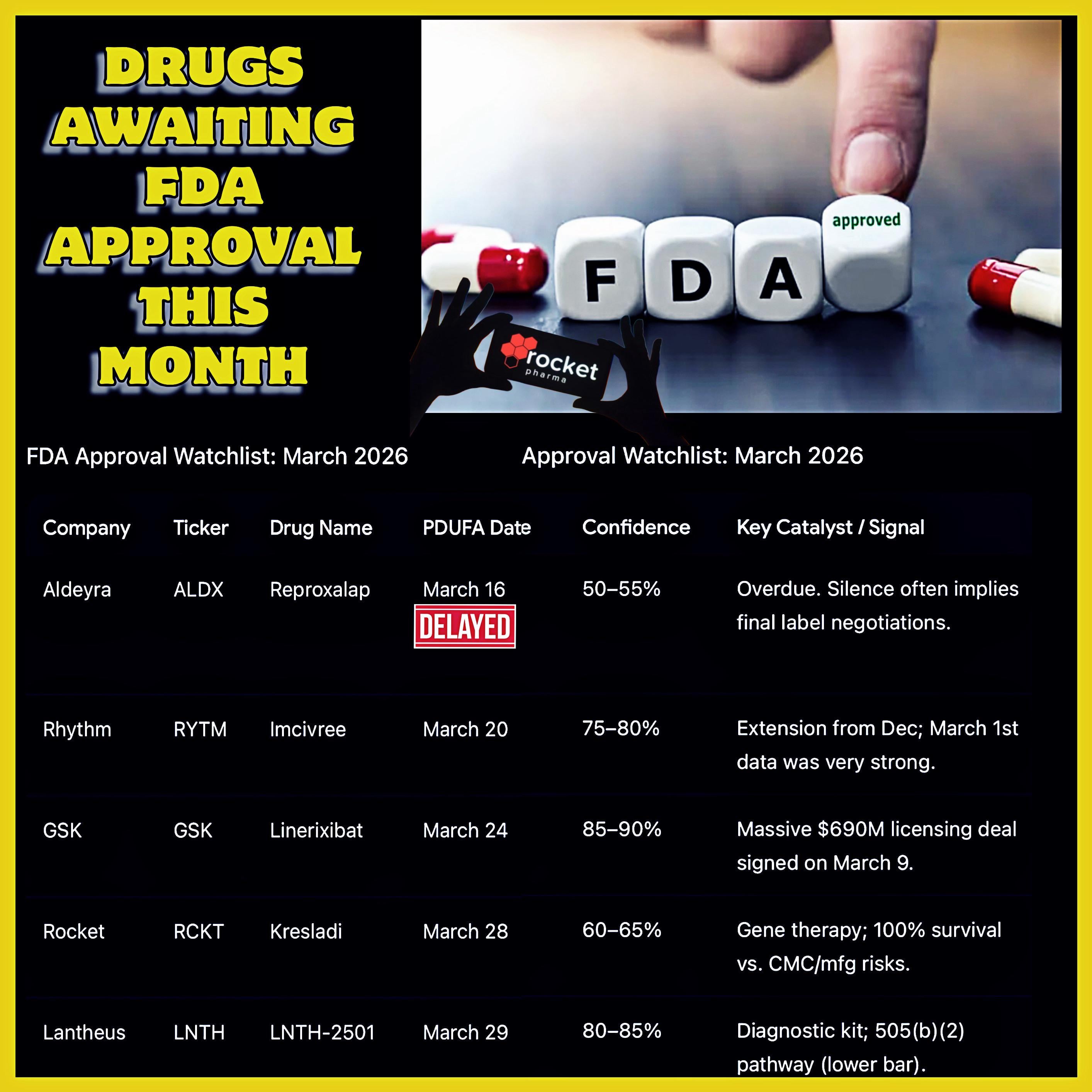
Dr. Donald Kohn has been developing gene therapies for rare pediatric immune disorders for over 30 years. His work has culminated in the first-ever U.S. Food and Drug Administration–approved therapy for severe leukocyte adhesion deficiency-I. According to MedicalXpress, this genetic condition is characterized by recurrent infections and often early death.
The FDA approval is a significant step forward in treating this rare immune disorder. As noted by the FDA, the approved gene therapy will provide new treatment options for patients with severe leukocyte adhesion deficiency-I. However, it is essential to understand the limitations of this approval and the potential risks associated with the therapy.
The clinical trial that led to the FDA approval was a crucial step in the development of this gene therapy. As reported by ClinicalTrials.gov, the trial demonstrated the safety and efficacy of the therapy in treating severe leukocyte adhesion deficiency-I. However, the exact details of the clinical trial and the gene therapy are not specified in the source, highlighting the need for further research and transparency.
soft editorial photography, diffused natural window light, cool neutral overcast light, flat even illumination. A close-up detail or consequence📷 Photo by Tech&Space
A significant step forward in treating rare immune disorders
The implications of this approval are significant, with potential benefits for patients with severe leukocyte adhesion deficiency-I. As stated by the National Institutes of Health, gene therapies hold promise for treating rare and debilitating diseases. However, it is crucial to approach this development with caution and consider the potential risks and limitations.
The FDA approval is a testament to the progress being made in the field of gene therapy. As noted by ScienceDaily, researchers are making significant strides in developing new treatments for rare and complex diseases. The approval of this gene therapy is a step forward, but it is essential to continue researching and developing new treatments to address the needs of patients with severe leukocyte adhesion deficiency-I.
The future of gene therapy holds much promise, but it is crucial to approach this field with caution and consider the potential risks and limitations. As reported by Nature, researchers are working to develop new gene therapies that can address a range of rare and debilitating diseases. However, further research is needed to fully understand the potential benefits and risks of these therapies.
This development has significant implications for future research and patient care, with potential benefits for patients with severe leukocyte adhesion deficiency-I. The approval of this gene therapy is a step forward, but it is essential to continue researching and developing new treatments to address the needs of patients with rare and complex diseases. For all the noise, the actual story is that gene therapies hold promise, but further research is needed to fully understand the potential benefits and risks.