Whole-genome sequencing delivers diagnoses—with limits

Whole-genome sequencing delivers diagnoses—with limits📷 Published: Apr 14, 2026 at 12:21 UTC
- ★23% diagnosis rate in 15,000 rare-disease genomes
- ★Routine sequencing at Karolinska, but not everywhere
- ★Study flags gaps in genetic interpretation tools
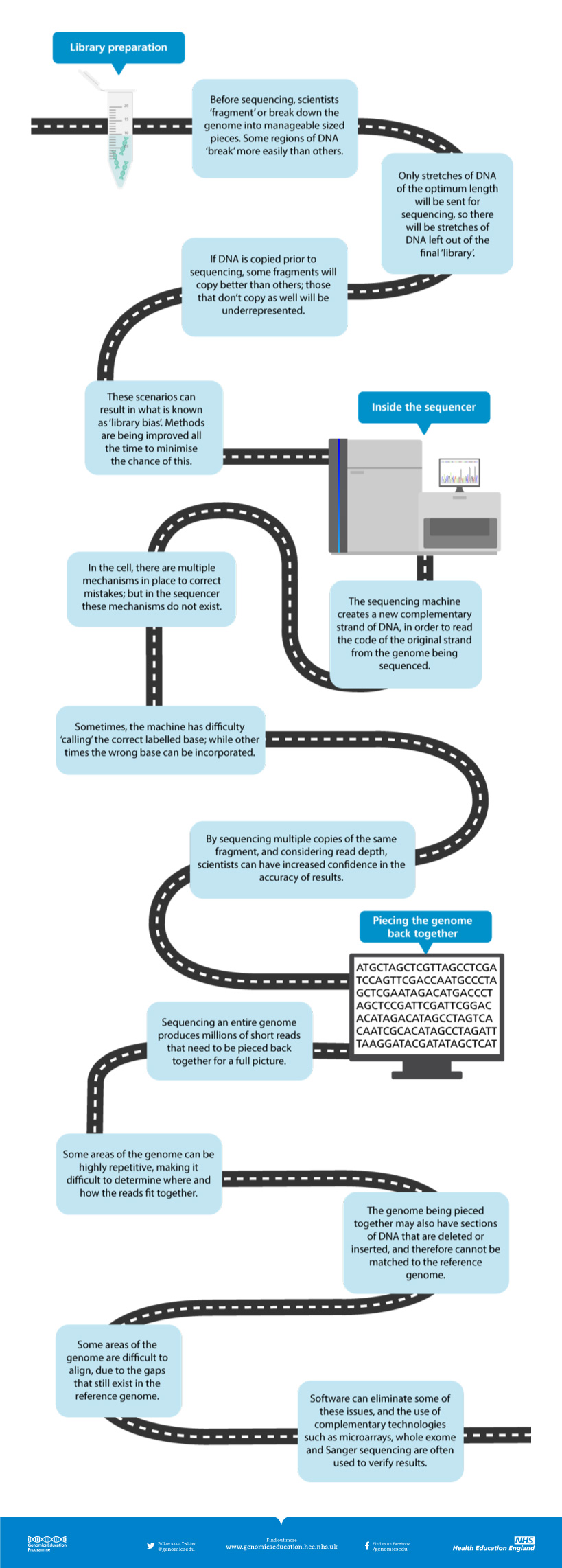
The integration of whole-genome sequencing (WGS) into routine diagnostics for rare diseases is no longer theoretical. A collaboration between Karolinska Institutet, Karolinska University Hospital, and SciLifeLab has sequenced the genomes of more than 15,000 patients, yielding a genetic diagnosis for 23%—a rate that aligns with earlier observational studies but remains far from universal. Published in Genome Medicine, the findings confirm that WGS can identify elusive genetic causes, but they also reveal the technology’s current ceiling: most patients still leave without answers.
The study’s design matters. Unlike targeted gene panels, WGS scans all 3 billion base pairs, increasing the odds of spotting rare or novel variants. Yet even this exhaustive approach delivered diagnoses for fewer than one in four patients. The remaining 77% highlight a critical bottleneck: not all genetic variations are well-understood, and not all can be linked to disease with today’s tools.
This isn’t a failure of sequencing—it’s a limit of interpretation. The study authors emphasize that while WGS expands diagnostic reach, its effectiveness hinges on reference databases, clinical expertise, and the inherent complexity of rare diseases, many of which involve multiple genes or non-coding regions still poorly characterized.
A large study—with real limits📷 Published: Apr 14, 2026 at 12:21 UTC
A large study—with real limits
For patients, the implications are cautiously positive. A 23% diagnosis rate means thousands now have a molecular explanation for symptoms that once defied classification—enabling tailored treatments, reproductive planning, or simply an end to diagnostic odysseys. But the flip side is stark: three-quarters still lack answers, and even a confirmed genetic cause doesn’t always translate to a therapy. Rare diseases often have no approved drugs, and WGS doesn’t bridge that gap.
The study also exposes disparities in access. Karolinska’s program is a model, but most hospitals worldwide lack the infrastructure for routine WGS. Cost, data storage, and the need for specialized genetic counselors remain barriers. Meanwhile, the 23% figure may overstate real-world performance; Karolinska is a tertiary referral center, meaning its patient pool is pre-filtered for likely genetic cases.
What’s missing from the headlines? The study doesn’t compare WGS to cheaper, faster methods like exome sequencing or gene panels—leaving open whether its higher diagnostic yield justifies the resource investment. And while the data are robust, they’re observational: no randomized trials here, just a snapshot of what happens when a well-funded system deploys cutting-edge tools.
In other words, this isn’t a breakthrough—it’s a benchmark. The 23% diagnosis rate reflects both the power of WGS and the vast unknowns that remain. For every patient who gets answers, three do not, and the study doesn’t explain why some variants are diagnosable while others aren’t.